Tuesday, 28 April 2026 — HART
MHRA are not telling anyone, but you can

Cheryl Grainger
Way back in 2022, Dame June Raine the now retired CEO of MHRA, did a presentation in Wales covering the 4 “pillars” of their safety monitoring. She mentioned that in the Yellow Card Vaccine MONITOR (YCVM), there were over 2,000 pregnant women and she was excited as this real-time data gave them a denominator, making the information more valuable.
….and then we heard nothing.
There is a special case for drug use in Pregnancy. Pregnancy should be significant to the MHRA. Thalidomide and the teratogenicity caused, forced governments and medical authorities to review their pharmaceutical licensing policies. The resulting 1968 Medicines Act, eventually lead to the formation of the MHRA in 2003. An important change was that drug trials for substances marketed to pregnant women, had to provide evidence that they were safe for use in pregnancy.
There was no safety data on the Covid-19 “vaccines” in pregnancy.
It was unprecedented that in 2021 a novel treatment (Covid-19 “vaccines”) had been rolled out to the pregnant, at the same time as those who were not pregnant.
MHRA describes the Monitor as:
- Real-time, targeted active monitoring of vaccinees.
- Enrolment prior to vaccination, recording of Adverse Drug Reactions (ADRs) at set intervals.
- Helping to estimate the frequency and long-term outcomes of side effects.
- A novel technology and methodology
MHRA states, “The YCVM has played a role in our strategy to rapidly detect, confirm, characterise and quantify new risks, communicating on these as necessary.” And yet on Page 2 of the report it says, “The primary purpose of the YCVM was not to detect safety signals or identify rare risks.”
The Monitor data should be more accurate than information randomly received via Yellow Card Reports.
- Yellow card data was produced every 2-weeks on the MHRA website – so why weren’t the more accurate Monitor results reported?
- How can it be right to take 4 years to publish the Monitor data, by its very nature more accurate than Yellow Card data, when it is showing very strong patterns of ADRs in high percentages of patients?
The method used was biased. Participants self-selected both as to whether they would get vaccinated AND whether they would volunteer for the survey. There is a lot in the research literature showing that these two things can individually and together bias a study and further biased by prioritising people of particular regions or ethnicities. This self-selection bias is “hidden” behind an untrue “we randomly selected them” claim.
They selected women of breeding age – so they were initially selected based on an immutable characteristic – and invited them via letter. Almost all the people, of which more than half are women, were registered and responding BEFORE the vaccine was available to pregnant women and two-thirds before it was generally available. Either the numbers tabulated are bad, or they are all healthcare workers who in this case come with an innate bias in favour of health treatment (vaccination).
The Monitor had run for 2 years till December 2022, and 36,604 participants had agreed to take part. Curious to know what the pregnancy data showed, I filed an FOI (23-280)in April 2023, asking the MHRA for the Monitor’s pregnancy data via a series of questions. The MHRA refused on the grounds of s22 which was their intension to peer review and publish (with no date given). This led me through the full FOI process ~ a laborious, inefficient system – that took two years.
Along with my refused FOI Request I was sent an Interim Report (July 2021). This was
a presentation by MHRA, concerned with drug safety to PEAG (Pharmacovigilance Expert Advisory Group), a sub-group of the CHM, concerned with risk assessment of medicines.
My FOIA Journey
Prof Norrman Fenton analysed this Interim Report and asked, “Why is the MHRA hiding critical safety data on the covid vaccines?” He called it scandalous. The MHRA seemed to say, “We have all the pregnancy outcome data, but we are not making it public yet”.
By April 2024, Prof Fenton stated, “the public are being misinformed as they are not being given up to date information as it is being withheld and when they find out the risks in pregnancy, they will be very concerned and annoyed they were not informed.”
In pregnancy we are warned about soft cheeses, alcohol etc and yet the MHRA have not shared safety data on novel, genetically engineered therapies.
The Interim report showed at least 1ADR was seen in 51% of the total and 41% of the pregnant. At the time, Yellow Card Reports in July 2021, were showing:
965,437ADRs (283,434 reports) with 1328 fatalities.
MHRA was still saying “safe and effective” and nearly all reactions were “expected acute reactions”.
If MHRA had read the Pfizer’s Common Technical Document, the data submitted for license, they would know Pfizer’s 90-day Post Marketing Report shows 1223 deaths by the end of February 2021.
The MHRA’s barrister worked the system because they weren’t ready, and the hearing was moved from September to December 2024. I was then informed the Preprint was to be published (13.11.24). I was told by the MHRA that my FOI questions had been answered and asked to withdraw; I was asked 4 times. I refused because the group of specialists I was working with told me the answers given could not be validated by the information in the Preprint.
I believed the MHRA has a duty of care to act in the Public Interest:
- to act on any data of harm
- to make information available to the public promptly
- to reduce the possibility of serious injury.
I made my arguments to continue, and the hearing was moved again to its final hearing slot of 20.2.2025. The Judge ordered us to complete the columns of a Scott Schedule: Column 1 = my questions
Column 2 = MHRA’s answers
Column 3 = My review of the missing data (30pages)
Column 4 = MHRA’s responses.
I did not have legal help at the hearing but was able to call on a generous lawyer giving his advice and time for free. When the judgement dismissed this case, I felt it was always going to be this way ~ “a fait accompli”. But importantly, I had forced them to release their report, so had sight of their interpretation of the data.
The judgement stated, “The MHRA and indeed the Commissioner had accepted that the public interest was weighted in favour of the release of the information, but this had to be balanced against the need to have proper analysis of all the data and a considered overview of safety. The publication of the raw data….could easily, as happened before, be misused, misquoted or misunderstood’.
“Misinformed hysteria about the vaccine may equally put lives at risk”.
The judgement was based on the need to have proper analysis but there was nothing proper about the analysis in the MHRA’s published report. The judgement seemed to be saying that the MHRA were the only group expert enough to analyse their own data!
“Implementation and results of active vaccine safety monitoring during the Covid-19 pandemic in the UK”
Wong, Donegan, Harrison, Jan, Cave, Tregunno
- All the 6 authors are MHRA employees.
- Cave (Chief Safey Officer), Tregunno (Patient Safety Monitoring).
- 86% MHRA funding provided by Pharma. No declared interests.
They finally published the paper in Drug Safety, Springer Nature Link in September 2025
MHRA’s Summary of their RESULTS
- 36,604 individuals registered, with 30,281 reporting vaccination.
No comparison to the unvaccinated.
- 15,764 (52.1%) reporting vaccination reported experiencing at least 1ADR.
- Nearly all were expected acute reactions. MHRA and its analysis tends to concentrate on pre-defined events of special interest = “observed versus expected”. Anything not seen before (VITT, turbo cancers, myocarditis), get
missed or take a very long time to surface.
- 4134 (13.7%) reported an event considered medically serious*.
- The data raised no safety concerns in pregnant and breastfeeding females.
- Reporting of menstrual disorders appeared stimulated by media interest, as seen in spontaneous reporting systems.
- Data on the incidence of tinnitus were used to support regulatory action on this signal.
*13.7% (1 in 7) had a MEDICALLY SERIOUS EVENT
MedRa scale for SERIOUS (5 categories): Death
Life-threatening
Hospitalisation
Disability or Permanent damage
Congenital or Birth defect
Prof Fenton and a group of specialists of highly qualified medics, scientists and analysts described the report and data as:
- The data does not substantiate MHRA’s statements
- The data highlights significant gaps in their information and statements
- No evidence of safety
- Evidence of harm especially in the pregnant
- Misleading
- Meaningless
- Underestimated
- Obfuscated
- Serious omissions
- Interpretation of data is inadequate
SUMMARY: Doesn’t answer the FOIA questions asked.
Can you believe the MHRA stated in their Preprint…………
“ONLY 13.7% (14,134 or 1 in 7) had a medically SERIOUS event”.
I cannot believe that the UK Regulator thinks it is acceptable to state “Only 13.7%” as if this is perfectly acceptable. “Only” was dropped on peer review and before publishing. This tells a great deal.
It seems, ‘1 in 7 experiencing a SERIOUS medical event’, is acceptable to the MHRA, the very people who are charged to look after pharmacovigilance (drug safety) on our behalf.
- How many pregnant women have been vaccinated whilst we waited 4years for these results to be published? We are talking about the % chance of damage to pregnant women, foetuses and babies.
- How many pregnant women will have been vaccinated whilst we wait for the MHRA to make us aware of the level of Serious ADR?
- Why are the MHRA not being transparent as they are withholding this information from pregnant women they are charged to protect?
Let us review the missing data and misinformation on pregnancy in the Monitor which negates the MHRA’s analysis. The missing and unreliable data provided by the MHRA:
- They only monitored pregnant women for an average of 73 Days (5-244)
10 weeks only – a full pregnancy is 40 weeks. Why did they leave the Monitor?
- Trimester information was missing and was not known in 762 (30%) of cases. Thalidomide showed its developmental effects if taken in the first trimester (day 34-50 after last period); the trimester is very important.
- In 2514 pregnancies, 106 spontaneous abortions would be expected~ only 19 were known in the Monitor.
- 8 Congenital anomalies are recorded but there is no detail given.
- No pre-term births were reported and only 4 premature births are mentioned, and this is in the Free Text reports. This is the only time Free Text reports are mentioned, and they are intentionally missing in the MHRA’s analysis.
- In the monitored breastfeeding women, 53.5% reported 900 ADRs.
- 1421 women were missing from the data.
They assumed non-respondents who didn’t report anything, didn’t have any ADRs.
- 50% of pregnancies were lost to follow-up – this is a critical failure. The protocol was to follow-up 10 weeks after birth. Follow-up data, particularly birth outcomes, is critical for understanding vaccine safety during pregnancy. We are talking about two lives here.
- MHRA’s conclusion: The data raised no safety concerns in pregnant and breastfeeding females.
The data and the way it is reported provides a misleading view of the safety of vaccines during pregnancy. Overall, there is an issue with outcome reporting bias, leading to misrepresentation of the risk-benefit ratio of vaccines. Concealing this information and suppressing public discourse is illegal. The MHRA continues to obscure data and avoid addressing gaps in their reporting. The MHRA does not adequately report or evaluate the data collected, which is a public authority duty.
A comparison can be made with the CDC’s V-Safe. We look at each country in isolation, as with the UK Covid Inquiry, instead of looking at the global picture.
The same monitoring system was used in the US, (V-Safe), but with 10million participants. Similarly to the MHRA, the CDC would not release the V-Safe data, but Informed Consent Action Network (ICAN) sued, and they were court ordered to release the data in September 2022. This was a release of more than 151million health surveys about experiences following Covid-19 vaccination.
The CDC did not publish an analysis, but ICAN did.
- The V-Safe data shows that 25% were incapacitated the day after vaccination.
- Overall, 7.7% (770,000) had a serious health event.
- Medical care was needed on average 2-3 times each after vaccination.
- Over 70% of participants required medical attention, emergency room intervention and/or hospitalisation.
Similarly, but worse, 97% (35,691) of the vaccinated pregnant women in the V-safe database and their babies had no outcomes recorded.
Again, this real time data is throwing up large numbers of ADRs, like the Interim Report and the YCVM data. This was not just recorded in the UK as it is a global problem. As with the MHRA, the CDC ignored these reports from its own V-safe program. The release of the data in the fourth V-Safe production reveals a disturbing trend in miscarriages and menstrual disturbances following covid-19 vaccination.
As reported, MHRA have not released or discussed the critical “free text” entries collected. The CDC were eventually court ordered by a federal judge, to release 7.8m free text entries from V-Safe. The CDC had sat on this information.
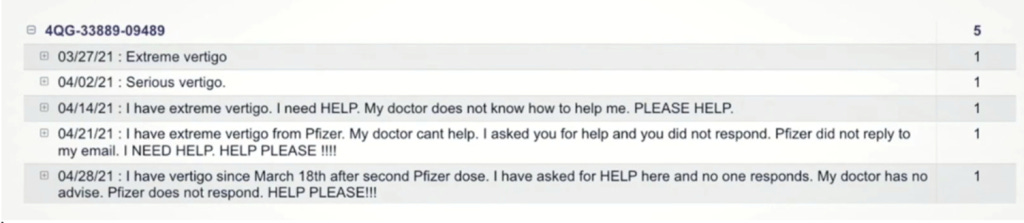
V-Safe free text reports were released via the Highwire and ICAN who obtained the data. Cries for help from a Vertigo Report and a Death Report:

SAFE & EFFECTIVE
The MHRA continues using the word “safe”. Dr Alison Cave, Chief Safety Officer MHRA, an author of the Monitor stated: “Patient safety is our highest priority. The Covid-19 vaccines were approved after a rigorous review of the safety, quality, and effectiveness of the vaccines by the MHRA and the CHM. The MHRA concluded that the Covid 19 vaccines were safe and effective, and the benefits of the vaccines outweigh any risks.” And this mantra has continued into its 5th year even though they have YCVM evidence of harm.
CONCLUSION
Given the well documented SAEs and unacceptable harm-to-reward ratio, governments should be urged to endorse and enforce a global moratorium on these modified mRNA products.
We should take great interest in the MHRA, who are responsible for the pharmacovigilance of the Covid–19 “vaccines”, a brand-new experimental technology. We should be particularly interested in the recording of information surrounding these liability-free vaccines. As members of the public, we should have substantial interest in reviewing the monitoring of these vaccines by the MHRA, introduced via an untested, accelerated review process.
I believe there is a very strong need for the public to be made aware of this available data and it should be used to update the advice given especially to those who are pregnant.
The MHRA are not telling the public the truth. I stepped forward to ask the MHRA important and intelligent questions. I believe that their answers are uninformed, and therefore, that they are not telling the public the truth. This occasions a lack of trust in their ability to protect the public from harmful substances. Lack of trust of the MHRA develops because of its duplicitous and disingenuous behaviour, demonstrated at its board meetings and appearances on other platforms. The MHRA has stated in board meetings that it believes it has the public’s trust.
Only through full and transparent disclosure of the complete data set can meaningful independent assessment take place. Without such disclosure, there remains a significant risk that the data could be biased, incomplete, or otherwise misinterpreted, potentially undermining public trust in the MHRA and its conclusions.
Do you believe the MHRA has demonstrated malfeasance in public office?
I conclude with a quote from the British Medical Journal, editorialised in January 2022:
“Pharmaceutical companies are reaping vast profits without adequate independent scrutiny of their scientific claims. The purpose of regulators is not to dance to the tune of rich global corporations and enrich them further; it is to protect the health of their populations. We need complete data transparency for all studies, we need it in the public interest, and we need it now”.
Leave a comment